SkinPen Remains the Only FDA-Cleared Device for Microneedling
Sponsored Content: Bellus Medical Aesthetic medical providers beware: The FDA [...]
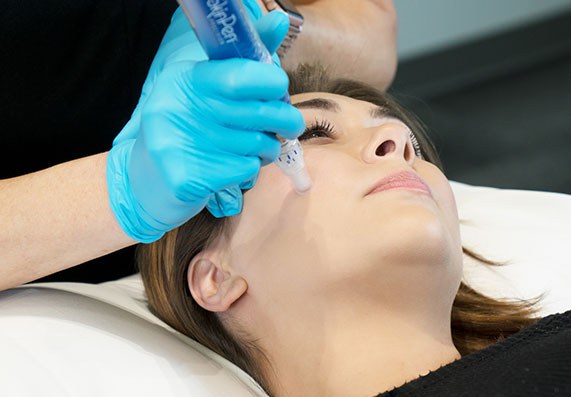
Sponsored Content: Bellus Medical
Aesthetic medical providers beware: The FDA codified its classification order for microneedling, likely putting practices using non-cleared devices at greater risk.
“In many medical device liability cases, a doctor is shielded from liability when properly using a cleared and compliant device, since the FDA review and clearance would ensure all likely hazards are controlled,” said Marc C. Sanchez, an FDA attorney who worked on Bellus Medical’s microneedling De Novo submission. “That defense is totally washed away when using an unapproved medical device.”
As of March 1, there is only one FDA-cleared microneedling device: SkinPen by Bellus Medical. According to Sanchez, the classification order makes all non-cleared microneedling devices of any kind subject to enforcement actions, such as refusing entry for imported derma rollers and pens as well as issuing warning letters to non-compliant manufacturers.
Read Sanchez’s post on FDA Atty to learn more about the classification order and the new risks for doctors and aestheticians.
Click here for more information on Bellus Medical, and click here to read more about the SkinPen in AmSpa’s medical spa treatment directory.
Share this article
Kate Harper
Follow us
An overview of the topics in this article.
Latest articles
March 17, 2026
March 17, 2026


