FDA Warns of Risk for Delayed-onset Inflammation Near Dermal Filler Sites
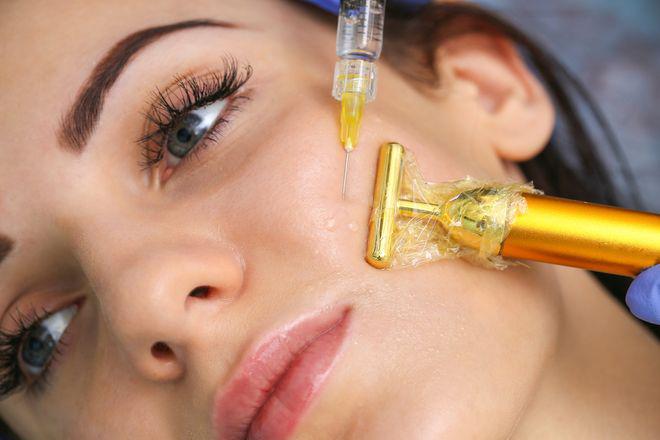
The US Food and Drug Administration updated its informational webpage on dermal fillers to reflect the risk of delayed-onset inflammation near dermal filler treatment sites.
The webpage now includes mention of delayed-onset inflammation near the dermal filler treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures along with risk of bruising, redness, swelling, and pain.
Typically, the reported inflammation is responsive to treatment or resolves on its own. The FDA encourages consumers and patients to discuss all treatment options, including dermal filler procedures, with their health care provider to understand the benefits and risks associated with the use of these medical devices, the FDA notes.
Read more at Modern Aesthetics >>