FDA Approves Topical Gel Filsuvez for Epidermolysis Bullosa
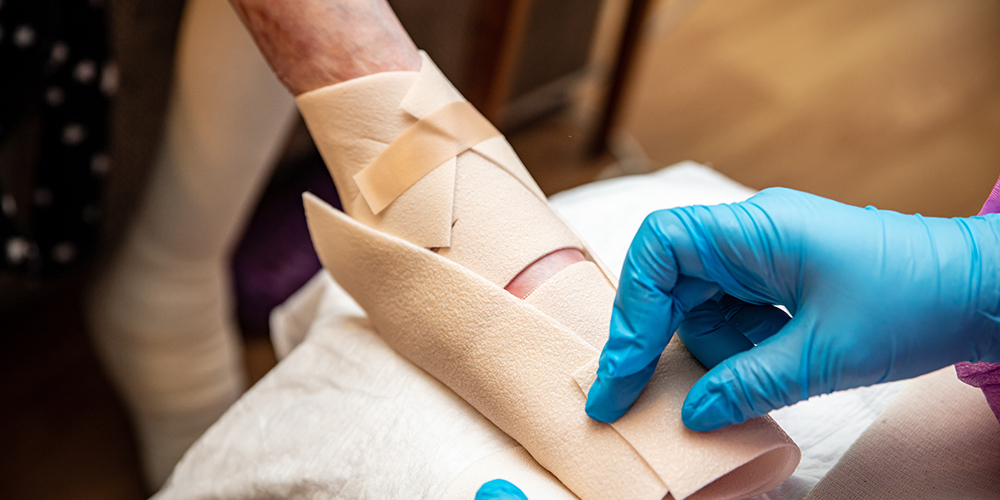
Chiesi Global Rare Disease recently announced the U.S. Food and Drug Administration’s (FDA) approval of Filsuvez (birch triterpenes) topical gel for the treatment of partial thickness wounds related to dystrophic junctional and epidermolysis bullosa (DEB; JEB) in patients six months and older.
The approval makes Filsuvez the first approved treatment for patients with wounds associated with JEB, a more rare and often severe form of EB that often begins with blisters notable in infancy.